“Phase 1: Where Science Becomes Medicine” was the apt title of last week’s conference in Manchester England, devoted to phase 1 trials in oncology. The ‘Science’ and ‘Medicine’ parts impressed me greatly. I’ll need another 2 weeks to follow up citations gleaned from the many excellent talks. The ‘Where’ was perfect, too—Manchester in the summer!
In this blog, though, I deal with the ‘Becomes’: translational methodologies that bridge preclinical science over into its clinical application as medicine. With dose finding long regarded as the prevailing methodologic concern in phase 1, the organizers could hardly have omitted a debate on the topic.
But the organizers missed a valuable opportunity when they conceived this debate as a mere technical squabble pitting one brand of 1-size-fits-all dose finding against another:
13:30 - 14:00 Question 1
Do “rules based” designs offer better value (in time, cost and identifying the recommended phase II dose range(s) than model-based designs?
Set up to argue the case for rules-based (aka ‘algorithmic’) 1-size-fits-all dose finding was S. Percy Ivy, MD, Associate Branch Chief of the NCI’s Investigational Drug Branch and Program Director for the Experimental Therapeutics Clinical Trials Network. Arguing for model-based 1-size-fits-all methods was Adrian Mander, Professor of Medical Statistics and Director of Statistics at Cardiff University.
Since learning of this planned debate early this year (before Dr Ivy replaced Jan Schellens on the programme) I had seen its far greater potential:
On Q1, if Jan Schellens exploits his first-mover advantage to highlight the 3+3/PC #DoseIndividualization design, with its demonstrable superiority against all conceivable 1-size-fits-all designs, will @Ademander have any choice but to flee the stage? 2/https://t.co/pIeZRELAUE
— David C. Norris, MD moved to Mastodon 🦣 (@davidcnorrismd) January 17, 2019
In late Feb, I emailed Dr Ivy to offer an amicus curiae brief in the form of an introduction to my Precautionary Coherence paper and its 3+3/PC design. We had a nice exchange that led me to think 3+3/PC had made a strong impression, and that Dr Ivy might indeed feature it in front of this important audience.
But alas, unseen diabolical forces went to work in the intervening months. In her slide deck, Dr Ivy acknowledged a handful of #OneSizeFitsAllogists for their assistance, with the usual proviso that she took responsibility for all errors. I would much rather blame the #OneSizeFitsAllogists.
In any case, a ‘debate’ that might have sizzled, fizzled. Dr Ivy dutifully trod through slides highlighting ideas from one camp of #OneSizeFitsAllogists, and Prof Mander highlighted the other half of his field. We got two bland, shambling presentations that I’ll admit at least had the merit of accurately mirroring the shambles that biostatisticians have made out of dose-finding. If the slides get posted, I’ll offer links here.
While the ‘debate’ itself hardly warrants comment, I do think that I managed to elicit some revealing responses from the debaters when I pressed the case for dose individualization from the audience mic:
“Good luck [with that]!”
Ouch! Number 4 on the list. These words came from a moderator of the debate, seemingly anxious to defend his debaters from the fairly withering criticism I’d just leveled. I’m no stranger to this defensive, squirming tone of dismissal. Still, when I hear it at a scientific conference, it always does surprise me just a little bit. After all, such cynicism clashes so profoundly with the spirit of intellectual enterprise and leadership that ostensibly prevails at such meetings.
“Regulators want a single dose”
As far as I can tell, this was the core of Dr Ivy’s objection to dose individualization—an all-too-familiar trope:
"The FDA made me do it…" is likely up there with "the dog ate my homework" for excuses about #clinicaltrials inefficiencies. Usually when I check the FDA denies that is true. Call out this excuse from pharma or @theNCI CROs. cc @US_FDA @FDAOncology @tmprowell @realrickpazdur
— Mike Thompson, MD, PhD, FASCO (@mtmdphd) August 6, 2018
When opponents of dose individualization retreat reflexively behind the regulatory ramparts this way, however, an optimist like me sees progress. Firstly, the retreat openly concedes the moral and technical case to me—since any person with a credible scientific or ethical argument would have led with that. Secondly, I am hardly convinced that today’s FDA is quite the impenetrable fortress against methodologic innovation that many would have us think. (If I’m wrong about this, then an August 12 FDA Workshop on Precision Dosing—just a few weeks away, as I write—should deliver plain evidence against me.)
Thirdly, as I pointed out from the mic, I have a ready counterexample: axitinib (Pfizer’s Inlyta), with an FDA label that includes 5 doses—2, 3, 5, 7 and 10 mg—plus an implicit dose-titration procedure:
“That’s just one drug”
Now with this retort, Dr Ivy almost reneges on the customary rules of logic, under which a counterexample is held to effectively refute a general claim. But for what it’s worth, I’ll offer here this second counterexample, a rather complex dose titration algorithm (DTA) for venetoclax:
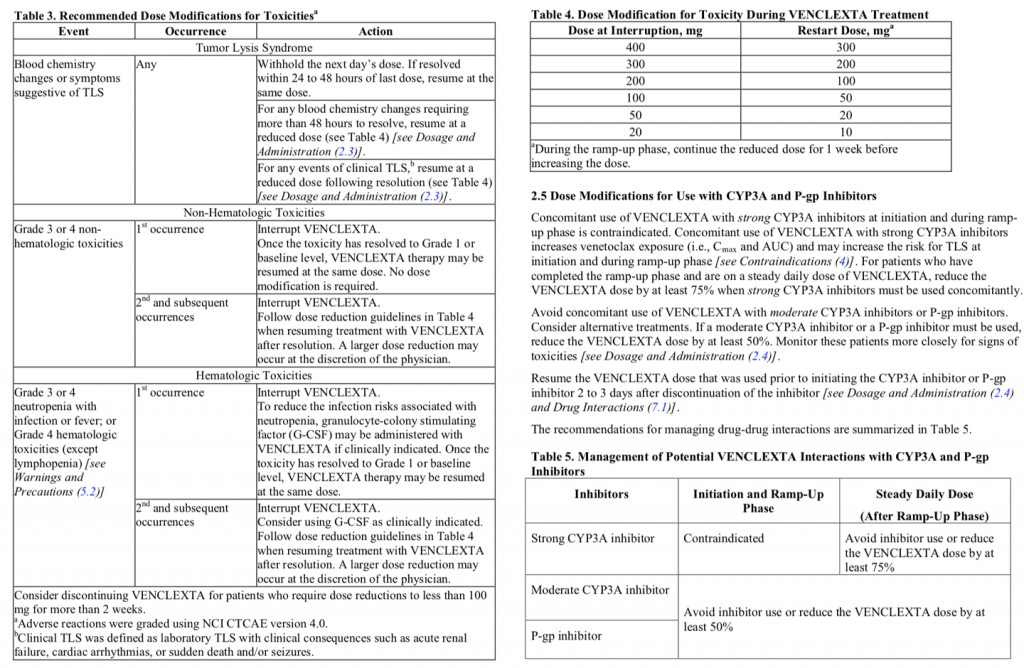
Source: https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/208573s000lbl.pdf
(Please, don’t anyone say, “But that’s only two counterexamples…”!)
“We already do dose adjustments”
This claim, again by Dr Ivy, is true enough. But again, if you just scratch its superficial veneer, this amounts to yet another frank concession to the dose individualization agenda. The very fact that phase 1 trialists find dose adjustments necessary—i.e., modifications to the 1-size-fits-all dose-finding designs biostatisticians impose on them—itself constitutes a powerful case for designing in dose individualization as a coherent first principle from the outset.
“They’re going to die, anyway.”
Wow. Even after surveying nearly 3 decades of 1-size-fits-all dose-finding methodology predicated on this atrocious view of phase 1 cancer trial participants, I was still shocked to hear Prof Mander say it out loud.
"They're going to die, anyway."
— David C. Norris, MD moved to Mastodon 🦣 (@davidcnorrismd) July 16, 2019
– @Ademander, in #Ph1MCR Session 9 explaining why phase 1 cancer trial participants don't deserve #DoseIndividualization.
The methodological content of 1-size-fits-all dose-finding designs, after all, objectively embodies precisely this view, through its utilitarian regard for participants. Still, it’s one thing to disguise such a view in mathematical formalism, and another entirely to state it in plain English. This degree of tone-deafness seems significant to me, for at least two reasons.
Firstly, although patient advocates did not attend this conference, they did have a substantial ‘presence’, through a series of sensitively produced video essays that opened each session. A recurring theme in these videos was the deep importance of hope as a value experienced by patients and their families through participation in phase 1 trials. In more than one of these videos, we heard from patients who had lived for many years on a trial: their hope, realized. Did Prof Mander see any of these videos? Did they leave any impression? On the contrary, what his remark reveals is the prevailing tendency within Biostatistics, to regard trial participants as remote abstractions. This is a point I addressed in my DTAT paper through Alfred North Whitehead’s notion of the fallacy of misplaced concreteness:

Secondly, we see in this remark just how endangered #OneSizeFitsAllipsism is, and how close to the edge of oblivion it is teetering. If even a single patient advocate had been in attendance at this debate, Prof Mander would have met with an immediately fatal rebuke. It is to the shame of every delegate at that conference—mine as well—that Prof Mander did not receive this immediate rebuke from one of us. That is the missed opportunity from Phase 1 Manchester that I most deeply regret.
